 
Always clean these loops with hydrochloric acid or nitric acid by dipping in it. You can use Platinum or nickel-chromium loops. There are two methods to perform the flame test. How to do a Flame Test?Īfter knowing what a flame test is? You must be wondering, how to do a flame test? Let us now look at the methods of performing flame tests. For example, the presence of Sodium turns the flame color to yellow. The flame test is a method used by scientists to observe the occurrence of specific metals in a compound by the color they give to a flame. 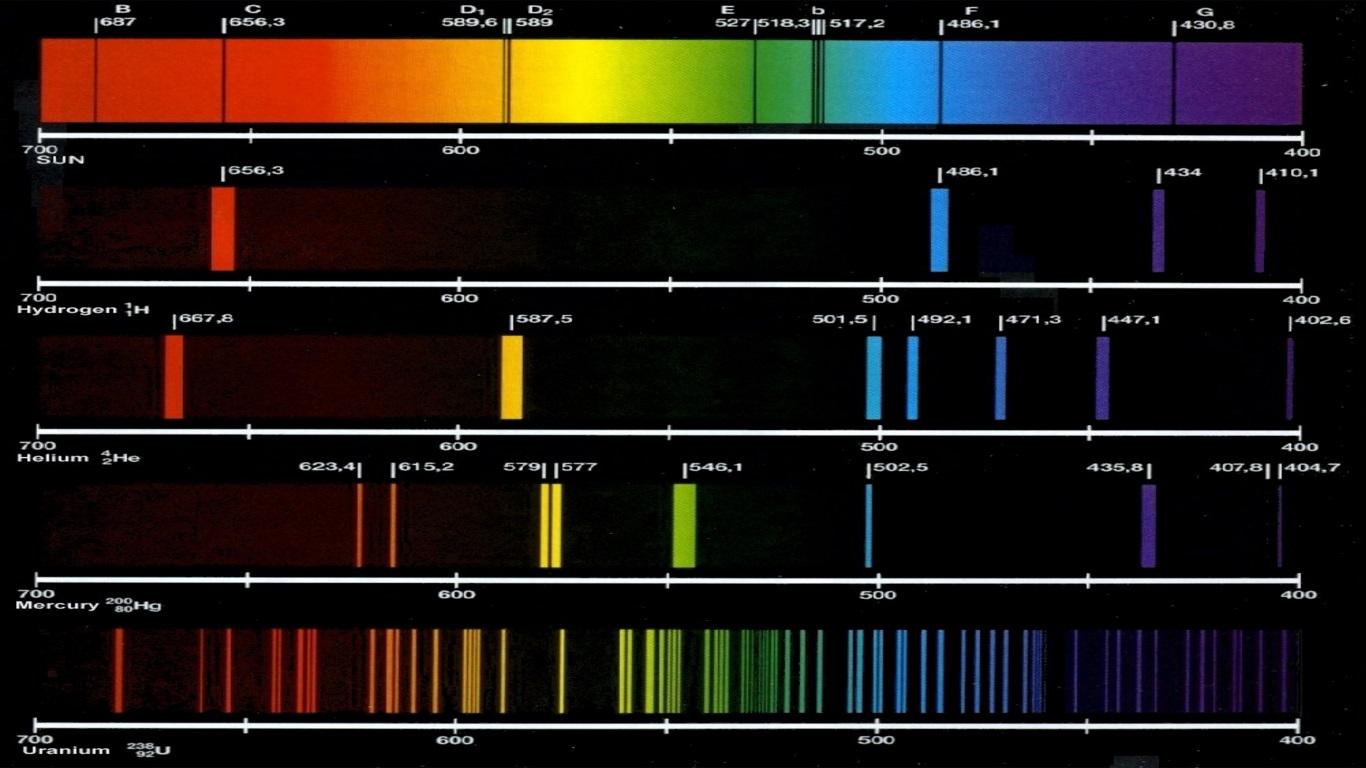 
Read the article below to know flame test definition in Chemistry, how to do a flame test, flame test color and limitation of the flame test. The transformation of electrons in the ions has a tendency to produce the visible color lines which are observed in flame tests. It should be observed here that the emission spectrum of each element that discovers the flame color includes atoms rather than ions. This difference is observed by the color of flames given out when the salt including the metal ion is burnt. The flame test for every element is different as ions of each element have a specific feature based on their emission spectrum. Generally, the flame test observes the occurrence of metal ions in a compound. It is primarily used to observe and analyze the presence of certain elements in the given compound or salt. The flame test is one of the most widely used analytical procedures in Chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
